
The rules listed are really meant to be general trends, which you may want to memorize. For example, although ionic compounds that contain halogens tend to be soluble, when the cation is lead (II) (as in lead (II) chloride or lead (II) iodide) the ionic compound will be insoluble in water. There are exceptions to the rules listed there. Notice that the title of the rules is general solubility rules. While the Ostwald step mle can be explained on the basis of nucleation kinetics, there is no thermodynamic contradiction in the initial formation of a finely divided precursor. 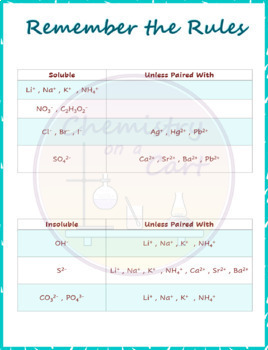
In other words, a supersaturated solution will nucleate first the least stable phase (often an amorphous solid phase) because its nucleation rate is larger than that of the more stable phase (Figure 13.26). The precipitation sequence results because the nucleation of a more soluble phase is kinetically favored over that of a less soluble phase because the more soluble phase has the lower solid- solution interfacial tension (7cw) than the less soluble phase (equation 50). This mle is very well documented mineral formation via precursors and intermediates can be explained by the kinetics of the nucleation process. The Ostwald step rule, or the mle of stages, postulates that the precipitate with the highest solubility (i.e., the least stable solid phase) will form first in a consecutive precipitation reaction. As an aid in predicting the products of precipitation reactions, some simple solubility rules are given in Table 4.1. 
Predicting the identity of the solid product in a precipitation reaction requires knowledge of the solubilities of common ionic substances. Precipitation reactions are examples of a double-displacement reaction. The solubility rules are used to determine if a product is insoluble. The molecular equation shows the reactants and products as molecules. 
These are given in all standard general chemistry texts, but they will be given here to aid in writing equations. To know if a precipitate will form as a product of a reaction in an aqueous solution, it is necessary to know a few simple solubility rules. Finally, you learned how to use the technique of titrations to determine the concentration of an acid or base solution. You learned about writing net ionic equations. You then learned about redox reactions and how to use an activity table to predict redox reactions. You examined the properties of acids and bases and the neutralization reactions that occur between them. Using a set of solubility rules allows you to predict whether or not precipitation will occur if two solutions are mixed. You also learned about electrolytes and nonelectrolytes. In this chapter, you learned about solutions and how to use molarity to express the concentration of solutions. You should apply these rules to all combination of cations with anions in each of the mixed solutions. To predict if precipitation will occur upon the mixing of two solutions, you must know and be able to apply the following solubility rules. Precipitation reactions involve the formation of an insoluble compound, a precipitate, from the mixing of two aqueous solutions containing soluble compounds. And remember-Practice, Practice, Practice. If you are unsure about mass/mole relationships, you may want to review Chapter 3. These rules will be useful as you learn to write net ionic equations. You may want to talk to your instructor and/or check your text for other solubility rules. We will present a set of solubility rules you can use to predict whether or not precipitation will take place when two solutions are mixed. Our goal in this chapter is to help you learn about reactions in aqueous solutions, including titrations. How would you use the solubility rules in Table 1.1 to separate the following pairs of ions In each case indicate what reagent you would add and write the net ionic equation for the precipitation reaction (a) lead(II) and copper(II) ions. The solubility rules in Table 1.1 are used to predict the outcomes of precipitation reactions. General rules on solubility help predict whether a solid-and what solid-will form when two solutions are mixed. Ī reaction where a solid forms is called a precipitation reaction. The precipitation sequence results because the nucleation of a more soluble. This rule is very well documented mineral formation via precursors and intermediates can be explained by the kinetics of the nucleation process. The Ostwald Step Rule, or the rule of stages postulates that the precipitate with the highest solubility, i.e., the least stable solid phase will form first in a consecutive precipitation reaction.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
